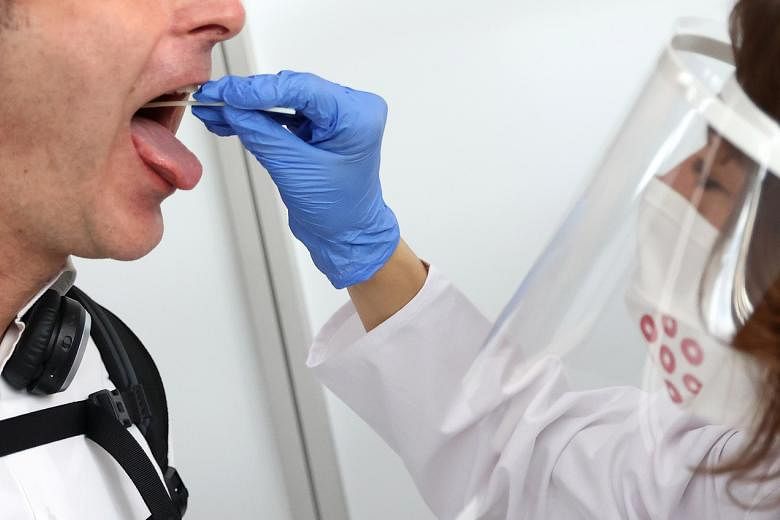
The use of saliva testing to detect Covid-19 is undergoing field tests in Singapore, as the health authorities assess if it can be scaled for widespread use.
Responding to queries from The Straits Times, the Ministry of Health (MOH) said on Thursday that it is assessing factors such as consistency and integrity of sample collection, efficiency of laboratory processes and the ability to cater for high volumes of tests via pooled testing.
It added that most samples for Covid-19 testing are collected through nasopharyngeal (back of the nose) or oropharyngeal (back of the throat) swabs, which can be pooled, thus allowing for high volumes of tests to be efficiently processed daily. Pooled tests involve using one Covid-19 test on a group of suspect cases, and if it yields a positive result, all individuals who were part of the group will be individually tested to determine the Covid-19 cases.
The MOH added that its preliminary findings showed saliva testing may be "slightly more comfortable for some individuals", though mechanisms to scale up testing in laboratories, including pooled testing, are still being developed.
In response to queries on why Singapore has taken longer in comparison with other countries to trial saliva testing, Professor Dale Fisher, a senior infectious diseases expert at the National University Hospital, said: "The lack of consistency in test collection methods may mean that it is difficult to standardise the collection process, in addition to the extra steps needed to process the tests in the lab, which could affect the labs' testing capacity.
"Before the tests can be scaled up, it is important for us to confirm that the saliva is sensitive enough as a sample, and that the labs can manage this very different specimen. Even then, I am not sure if any country does this as its mainstay of collecting specimens, but there would certainly not be many."
Associate Professor Raymond Lin, director of the National Public Health Laboratory at the National Centre for Infectious Diseases (NCID), said the centre is aware of three local studies done on saliva testing, all of which arrived at different conclusions, including an earlier study conducted by the centre.
"To validate various sampling methods, NCID is undertaking further study and analysis using a structured standardised testing approach with the approval from MOH," he said.
In a study led by Associate Professor Hsu Li Yang, programme leader of infectious diseases and co-director of global health at the National University of Singapore's Saw Swee Hock School of Public Health, it was shown that saliva testing through secretions taken from the back of the throat can be a viable method for Covid-19 testing.
The study, which has not been peer reviewed, took place from June 2 to June 26, and was conducted among 200 migrant workers who were recruited from community care facilities and dormitories.
They consisted of both confirmed and suspected cases, including those who were asymptomatic close contacts of confirmed cases, as well as patients who had symptoms of acute respiratory infection.
Three tests were done on the patients concurrently - nasopharyngeal swabs, saliva tests and self-administered nasal swabs - to compare their efficacy.
Of the three, the saliva test reflected the highest percentage of positive results, suggesting that it may be more sensitive than nasopharyngeal swabs, whereas self-administered nasal swabs were the least sensitive. As the swabs were self-administered, it is likely that they reached only the front part of the nasal passage, which has a lower viral load, therefore yielding poor results.
When asked about the possibility of false positives in the test results, Prof Hsu noted that false positives and negatives cannot be discounted entirely, as they occur in routine testing, but the chances of this occurring and the impact are very low.